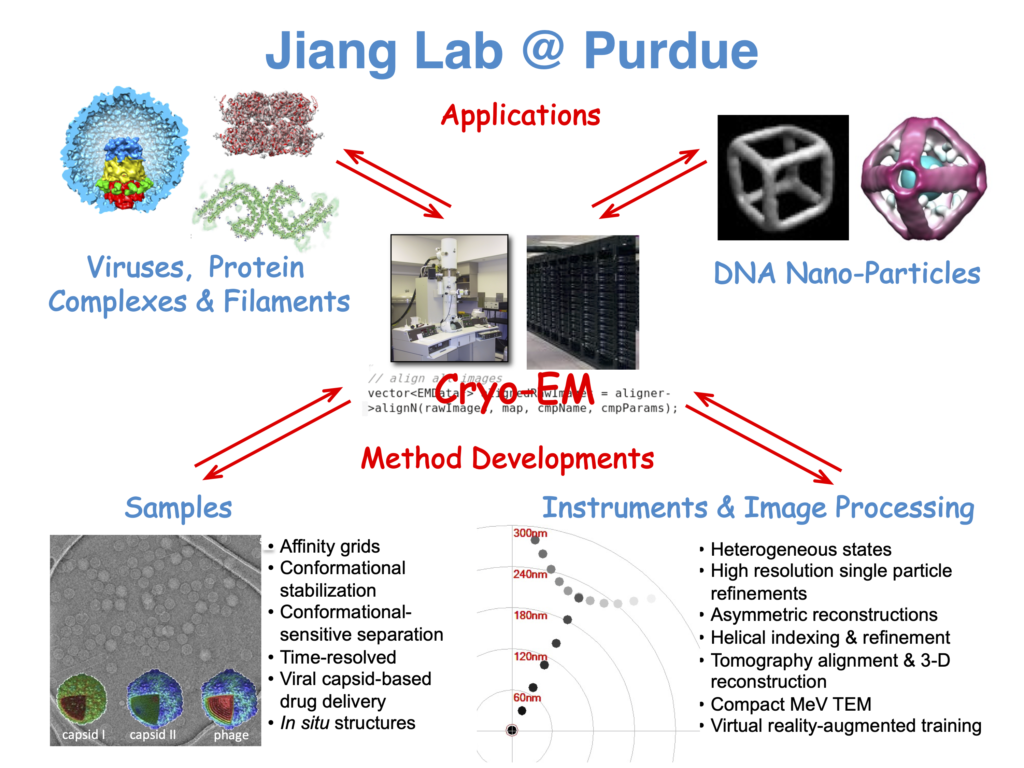
In the Jiang Lab we use cryo-electron microscopy (cryo-EM) to study structures of protein amyloids involved in neurodegenerative diseases, macromolecular complexes involved in cancers, human viruses, and bacteriophages. The Jiang Lab also develops methods to improve cryo-EM sample preparation, data collection, image analysis, and 3D reconstruction.










Follow Us